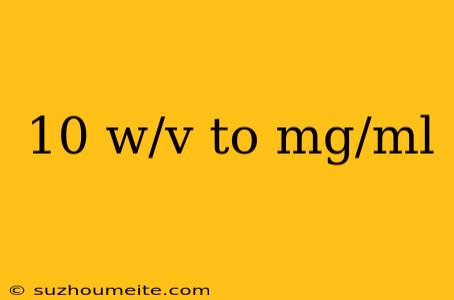10 w/v to mg/ml: A Comprehensive Guide to Concentration Conversion
When working with solutions, it's essential to understand the different ways to express concentration. Two common methods are weight/volume (w/v) and milligrams per milliliter (mg/mL). In this article, we'll explore how to convert between these two units, focusing on the conversion of 10 w/v to mg/mL.
What is w/v?
Weight/volume (w/v) is a unit of concentration that expresses the number of grams of a substance per 100 milliliters of solution. It's commonly used in pharmaceutical and scientific applications.
What is mg/mL?
Milligrams per milliliter (mg/mL) is a unit of concentration that expresses the number of milligrams of a substance per milliliter of solution. It's widely used in medical, scientific, and pharmaceutical applications.
Converting 10 w/v to mg/mL
To convert 10 w/v to mg/mL, we need to understand that 1% w/v is equivalent to 10 mg/mL. Therefore, we can convert 10 w/v as follows:
10 w/v = 10% w/v = 10 x 10 mg/mL (since 1% w/v = 10 mg/mL) = 100 mg/mL
Why is this conversion important?
Accurate conversion between w/v and mg/mL is crucial in various industries, including:
Pharmaceuticals
- Ensuring the correct dosage and concentration of medications
- Maintaining consistency in manufacturing processes
Research and Development
- Comparing results from different studies and experiments
- Ensuring accuracy in data analysis and interpretation
Healthcare
- Administering medications with precise dosages
- Maintaining patient safety and well-being
Conclusion
In conclusion, converting 10 w/v to mg/mL is a straightforward process that requires understanding the equivalence of 1% w/v to 10 mg/mL. By applying this conversion, we can ensure accuracy and consistency in various industries where concentration is critical. Remember, 10 w/v is equivalent to 100 mg/mL.