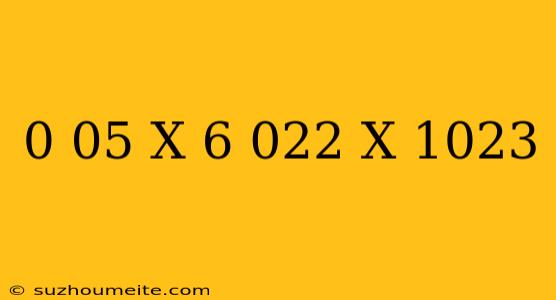The Magic of Avogadro's Number
Have you ever wondered what happens when you multiply 0.05 by 6.022 x 10^23? It may seem like a random combination of numbers, but trust us, it's a calculation that holds a special significance in the world of chemistry.
What is Avogadro's Number?
Avogadro's Number, denoted by NA, is a fundamental constant in chemistry that represents the number of particles (atoms or molecules) in one mole of a substance. It was named after the Italian scientist Amedeo Avogadro, who first proposed the concept in the 19th century.
The value of Avogadro's Number is approximately 6.022 x 10^23 particles per mole. This number is a crucial constant in many chemical calculations, including the calculation of molecular weights, reaction stoichiometry, and gas volumes.
The Calculation: 0.05 x 6.022 x 10^23
So, what happens when we multiply 0.05 by Avogadro's Number?
0.05 x 6.022 x 10^23 = 3.011 x 10^22
This calculation represents the number of particles in 0.05 moles of a substance. To put this number into perspective, 3.011 x 10^22 is an enormous quantity, equivalent to the number of stars in the observable universe!
Real-World Applications
Avogadro's Number has numerous applications in various fields, including:
Chemistry
- Calculating molecular weights and molar volumes
- Determining reaction stoichiometry and yield
- Analyzing chemical composition and structure
Physics
- Calculating the ideal gas constant (R)
- Determining the Boltzmann constant (kB)
Engineering
- Designing chemical reactors and process systems
- Optimizing chemical reactions and processes
Biology
- Studying the structure and function of biomolecules
- Analyzing the chemical composition of biological systems
Conclusion
In conclusion, the calculation 0.05 x 6.022 x 10^23 may seem like a simple arithmetic operation, but it represents a fundamental concept in chemistry that has far-reaching implications in various fields. Avogadro's Number is a powerful tool that helps us understand the behavior of particles at the molecular level, and its applications continue to shape our understanding of the world around us.