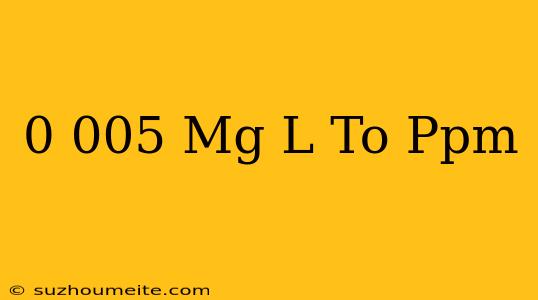Convert 0.005 mg/l to ppm
In chemistry, the concentration of a solution can be expressed in various units, including milligrams per liter (mg/l) and parts per million (ppm). While both units represent the amount of a substance per unit volume, they are not interchangeable. In this article, we will explore how to convert 0.005 mg/l to ppm.
Understanding the Units
Milligrams per Liter (mg/l)
Milligrams per liter is a unit of concentration that represents the number of milligrams of a substance present in one liter of a solution. It is commonly used in chemistry and environmental science to express the concentration of pollutants or contaminants in water or air.
Parts per Million (ppm)
Parts per million is a unit of concentration that represents the number of parts of a substance per million parts of a solution. It is commonly used in chemistry, biology, and engineering to express the concentration of a substance in a solution.
Conversion Factor
To convert mg/l to ppm, we need to know the conversion factor. The conversion factor is based on the density of water, which is approximately 1 gram per milliliter (g/mL). Since there are 1,000 milliliters in a liter, the conversion factor is:
1 mg/l = 1 ppm
Converting 0.005 mg/l to ppm
Now that we have the conversion factor, we can convert 0.005 mg/l to ppm.
0.005 mg/l = 0.005 ppm
Therefore, 0.005 mg/l is equivalent to 0.005 ppm.
Conclusion
In conclusion, converting mg/l to ppm is a simple process that requires a basic understanding of the units and the conversion factor. By applying the conversion factor, we can easily convert 0.005 mg/l to 0.005 ppm. This conversion is essential in various fields, including chemistry, biology, and environmental science, where accurate concentration measurements are critical.